Gets to work immediately by continuously releasing medicine into your eye.*

Maintains your vision.†‡

The only twice-yearly treatment for wet AMD.*

Ranibizumab, the medicine inside the implant, has been trusted by retina specialists for years.
If your retina specialist determines that it is necessary, you may need an anti-VEGF injection in the gel-like part of the SUSVIMO-treated eye (intravitreal) before your next refill. Only 1.6% of patients received additional treatment in the first 6 months. In the following 6 months, 5.4% of patients received additional treatment.
†In a clinical trial, 248 people received SUSVIMO, while 167 people were treated with monthly ranibizumab injections. On average, people on SUSVIMO were able to maintain similar vision results to those receiving monthly ranibizumab injections over 1 year.
‡There was a temporary decrease in vision following the SUSVIMO procedure; however, your vision will return to normal as your eye recovers from the procedure.
There is more to know about the implant insertion and refill procedure, refer to the SUSVIMO Implant Procedure for more information and procedure guidance.
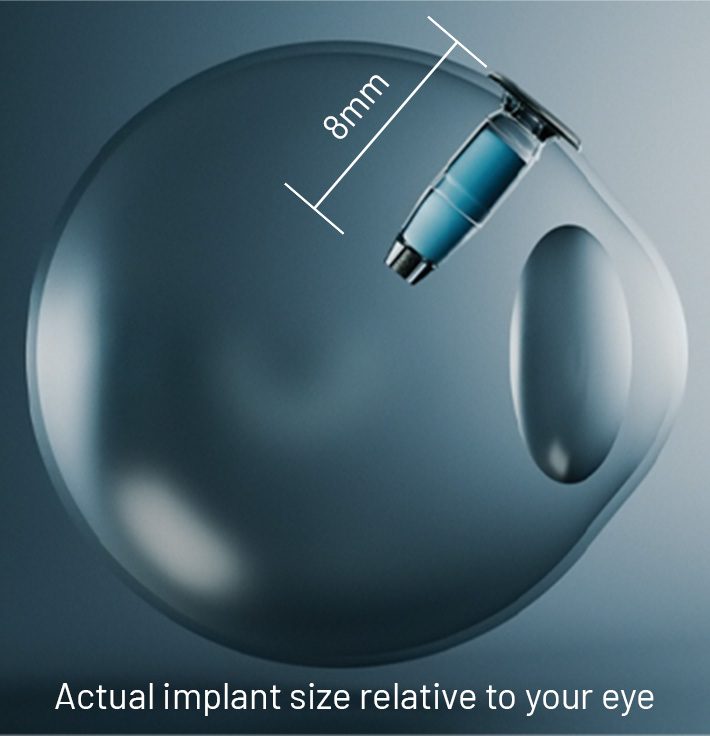
SUSVIMO is 8mm long, about the size of a grain of rice.
SUSVIMO is 8mm long, about the size of a grain of rice.
After it is implanted, you can’t feel it and others won’t see it in your eye.
LOOKING FOR RESOURCES?
LOOKING FOR RESOURCES?
This downloadable brochure can help to keep information close by about how SUSVIMO can help you get the most out of your treatment.
-
-
SUSVIMO [package insert]. South San Francisco, CA: Genentech, Inc; 2022.
SUSVIMO [package insert]. South San Francisco, CA: Genentech, Inc; 2022.
-
Campochiaro PA, Marcus DM, Awh CC, et al. The port delivery system with ranibizumab for neovascular age-related macular degeneration. Ophthalmology. 2019;126:1141-1154.
Campochiaro PA, Marcus DM, Awh CC, et al. The port delivery system with ranibizumab for neovascular age-related macular degeneration. Ophthalmology. 2019;126:1141-1154.
-
SUSVIMO Initial Fill and Implant Procedure Instructions for Use. Genentech, Inc. 2022.
SUSVIMO Initial Fill and Implant Procedure Instructions for Use. Genentech, Inc. 2022.
-
Monés J, Gune S, Maia M, et al. Pharmacokinetic profile of the port delivery system with ranibizumab in the phase 3 Archway trial. Presented at 21st EURETINA Congress, September 9-12, 2021.
Monés J, Gune S, Maia M, et al. Pharmacokinetic profile of the port delivery system with ranibizumab in the phase 3 Archway trial. Presented at 21st EURETINA Congress, September 9-12, 2021.
-
Ranade SV, Wieland MR, Tam T, et al. The port delivery system with ranibizumab: a new paradigm for long-acting retinal drug delivery. Drug Delivery. 2022;29(1):1326-1334.
Ranade SV, Wieland MR, Tam T, et al. The port delivery system with ranibizumab: a new paradigm for long-acting retinal drug delivery. Drug Delivery. 2022;29(1):1326-1334.
-
Data on file. Genentech, Inc. 2021.
Data on file. Genentech, Inc. 2021.
-
Data on file. Genentech, Inc. 2021.
Data on file. Genentech, Inc. 2021.
-
Holekamp NM, Campochiaro PA, Chang MA, et al; Archway Investigators. Archway randomized phase 3 trial of the port delivery system with ranibizumab for neovascular age-related macular degeneration. Ophthalmology. 2022;129(3):295-307.
Holekamp NM, Campochiaro PA, Chang MA, et al; Archway Investigators. Archway randomized phase 3 trial of the port delivery system with ranibizumab for neovascular age-related macular degeneration. Ophthalmology. 2022;129(3):295-307.
-
A phase III study to evaluate the port delivery system with ranibizumab compared with monthly ranibizumab injections in participants with wet age-related macular degeneration. ClinicalTrials.gov identifier: NCT03677934. Updated July 22, 2021. https://clinicaltrials.gov/ct2/show/NCT03677934
A phase III study to evaluate the port delivery system with ranibizumab compared with monthly ranibizumab injections in participants with wet age-related macular degeneration. ClinicalTrials.gov identifier: NCT03677934. Updated July 22, 2021. https://clinicaltrials.gov/ct2/show/NCT03677934
-
Wykoff CC; Archway Investigators. 2-year outcomes from the phase 3 Archway trial: management of neovascular age-related macular degeneration using the port delivery system with ranibizumab (PDS). Presented at Bascom Palmer Eye Institute Angiogenesis, Exudation, and Degeneration 2022 – Virtual Edition, February 11-12, 2022.
Wykoff CC; Archway Investigators. 2-year outcomes from the phase 3 Archway trial: management of neovascular age-related macular degeneration using the port delivery system with ranibizumab (PDS). Presented at Bascom Palmer Eye Institute Angiogenesis, Exudation, and Degeneration 2022 – Virtual Edition, February 11-12, 2022.
-
Data on file. Genentech, Inc. 2021.
Data on file. Genentech, Inc. 2021.
-
Data on file. Genentech, Inc. 2021.
Data on file. Genentech, Inc. 2021.
-
Chang MA, Kapre A, Kaufman D, et al. Patient preference and treatment satisfaction with a port delivery system for ranibizumab vs intravitreal injections in patients with neovascular age-related macular degeneration: a randomized clinical trial. JAMA Ophthalmology. Published online June 16, 2022.
Chang MA, Kapre A, Kaufman D, et al. Patient preference and treatment satisfaction with a port delivery system for ranibizumab vs intravitreal injections in patients with neovascular age-related macular degeneration: a randomized clinical trial. JAMA Ophthalmology. Published online June 16, 2022.
-
-